
banner
Time of issue:2019-07-09 00:00:00
All categories
Technology
- Categories:Our science
- Time of issue:2019-05-29 00:00:00
- Views:0
Description:
Information
1.More than 500 cases of exploratory clinical research have been completed in hematological tumor, and the clinical efficacy of The CAR-T products of Bio-raid is better than international.
(1) CD19+CD22 CAR-T cell sequential therapy
The first CD19+CD22 CAR-T cell sequential therapy can target CD19 and CD22, respectively, to avoid the recurrence caused by the escape mechanism of cancer cells under the pressure of single-target CAR-T cell therapy. The clinical efficacy is better than international.
(2) CD19/22 Dual-CAR-T
CD19/22 Dual-CAR-T treatment of diffuse large B lymphoma which was proposed firstly, can target CD19 and CD22 simultaneously, to avoid tumor cell escape.
(3) PD1 knockdown CAR-T
The shRNA that can knock down PD1 can reduce the expression of PD1 on the surface of CAR-T cells, thereby reducing the inhibition of PD-L1 on CAR-T cells in the tumor microenvironment and improving the anti-tumor effect of CAR-T cells.
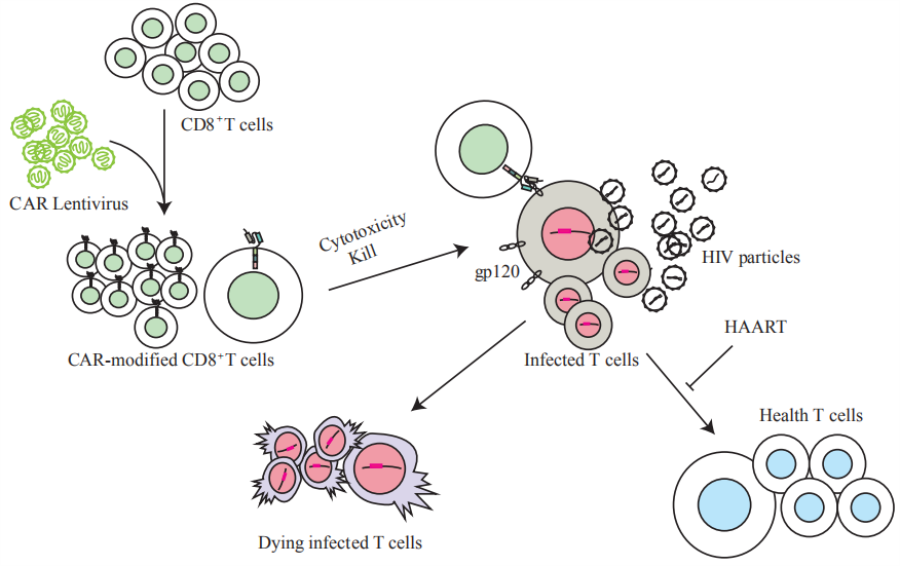
2. Viral infectious disease CAR-T treatment
Bio-Raid Inc. is the first company in the world to conduct HIV CAR-T clinical trial research, the technology of GP120 CAR-T treated infectious HIV has been authorized by the State Intellectual Property Office, and the PCT (Patent Cooperation Treaty) has entered the national review stage.
The advantage of our technology lies in the design of the new CAR molecule:
(1) Using a broad-spectrum neutralizing antibody that highly combined to the viral proteins Gp120 and Gp41 as scFv, the CAR overcomes the shortcomings of early design, enabling it to bind to most existing HIV-1 strains and improve its clinical adaptability.
(2) While using a safety-increased SIN (Self-inactivating) structure of the lentiviral vector, the CAR molecule is intracellularly engineered to be a double-stimulated to enhance cell expansion and survival, and to increase clinical efficacy and safety.
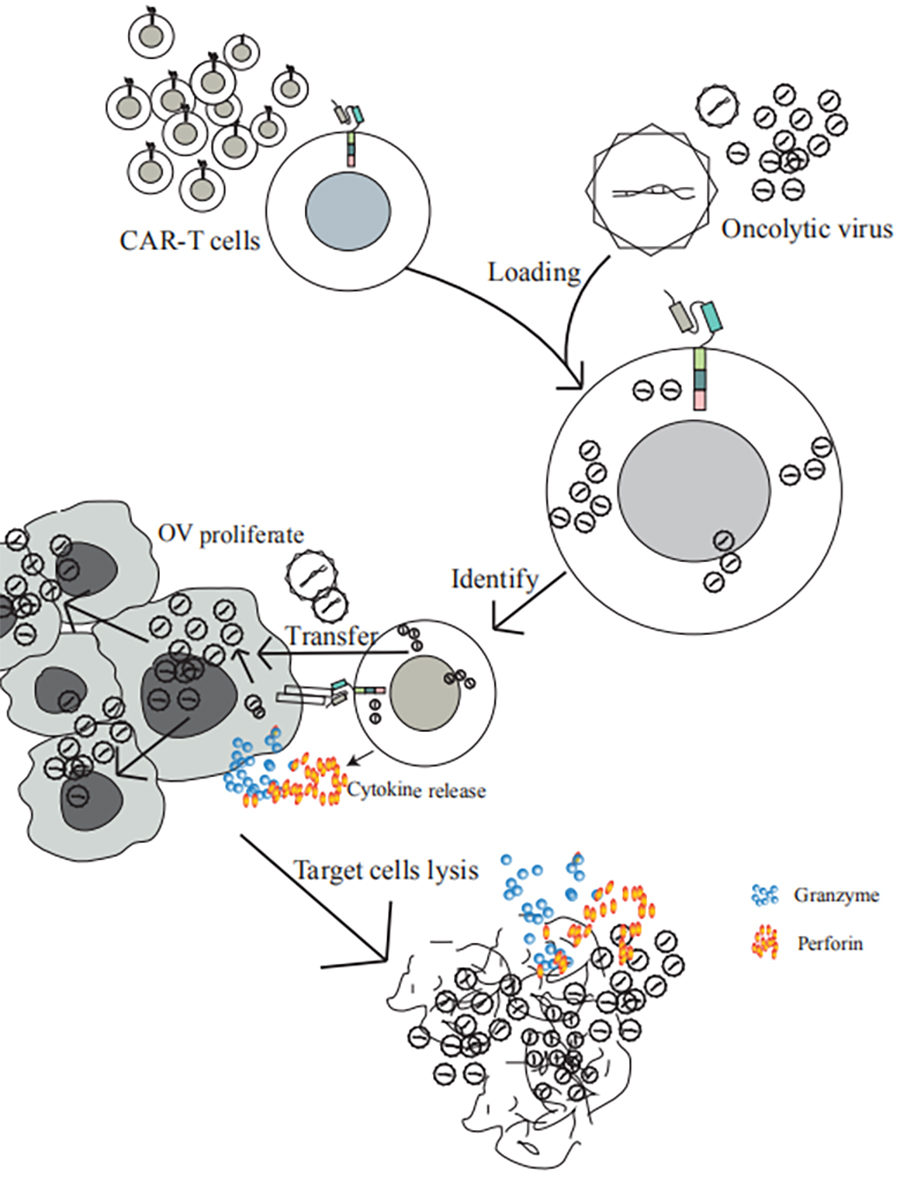
3. The patent that CAR-T combined with oncolytic virus for the treatment of solid tumor has been in the national trial stage.
This technology that CAR-T cells are modified for transporting the oncolytic virus to the solid tumors sites as oncolytic virus carriers, not only overcomes the limitations of CAR-T cells against solid tumors, but prevents the oncolytic virus from complicated humoral immune environment. The oncolytic virus are specifically carried and delivered to the solid tumors sites according to the specific targets of solid tumors by the CAR-T cells, and then the robust synergistic ability of CAR-T and oncolytic virus eliminating solid tumors is fully exerted.
4. The Universal CAR-T patent has entered the substantive examination stage.
The advantages of our universal CAR-T technology are as follows:
(1) Raw materials are the blood of healthy donors, which overcoming the problem that patients' T cells are difficult to obtain or cannot be used as raw materials for production (such as HIV patients);
(2) Instead of a custom-made cell therapy, Universal CAR-T can be supplied off-the-shelf , shortening the time for preparing drugs for patients;
(3) Knock out TCR B2M related genes by CRISPR gene editing technology to avoid graft-versus-host disease (GVHD) and minimize its immunogenicity, and provide timely and repeated reinfusion treatment for more patients;
(4) The PD-1 immune checkpoint was knocked down to further enhance the function of CAR-T cells.
The company currently has more than 6,000 square meters of scientific research base and the pilot plant. The GMP level of pilot plant were built mainly including plasmid production workshop, production workshop, cell production workshop, the physical and chemical lab through the introduction of high-tech equipment. Synchronously, to comply with GMP standards, standardized operation procedures and quality systems have been developed. At present, Bio-Raid is engaging on the independent research and development of a variety of CAR-T immune cell therapy drugs, such as hematologic tumors, viral infectious diseases (HIV, etc.), solid tumors and universal CAR-T. We have developed 14 CAR-T immunocytotherapies which target CD19, CD20, CD22, CD30, CD33, BCMA, GP120, EGFR/EGFRVIII, Mesothelin etc. Currently, many of them have entered exploratory clinical trials, and two of them are under IND application.
Scan the QR code to read on your phone

Wuhan Bio-Raid Biotechnology Co., Ltd.
Company address : 388 gaoxin 2nd road, east lake new technology development zone, wuhan city Floor 4, building 3, phase 3.1, wuhan optical valley international bio-pharmaceutical enterprise accelerator Floor 5-1
Phone number : 086-027-59318269
E-mail:cart@bio-raid.com

Contact
© Wuhan Bio-Raid Biotechnology Co.,Ltd. www.300.cn 鄂ICP备16000510号-1
